
CHANGING THE
PATH IN HAE1,2
CHANGING THE
PATH IN HAE1,2
THE FIRST AND ONLY
FXIIa INHIBITOR FOR HAE1,2
ANDEMBRY® (garadacimab) is indicated for routine prevention of recurrent attacks of hereditary angioedema (HAE) in adult and adolescent patients aged 12 years and older. 1
The person represented in this material is not an actual patient.

NOW NICE APPROVED3
NOW NICE APPROVED3

A DIFFERENT WAY
TO TARGET HAE1,2
ANDEMBRY® IS A FIRST-IN-CLASS, FULLY HUMAN,
ANTI-FXIIa MONOCLONAL ANTIBODY THAT
INHIBITS THE CATALYTIC ACTIVITY OF FXIIa1,2
ANDEMBRY® IS A FIRST-IN-CLASS, FULLY HUMAN, ANTI-FXIIa MONOCLONAL ANTIBODY THAT INHIBITS THE CATALYTIC ACTIVITY OF FXIIa1,2

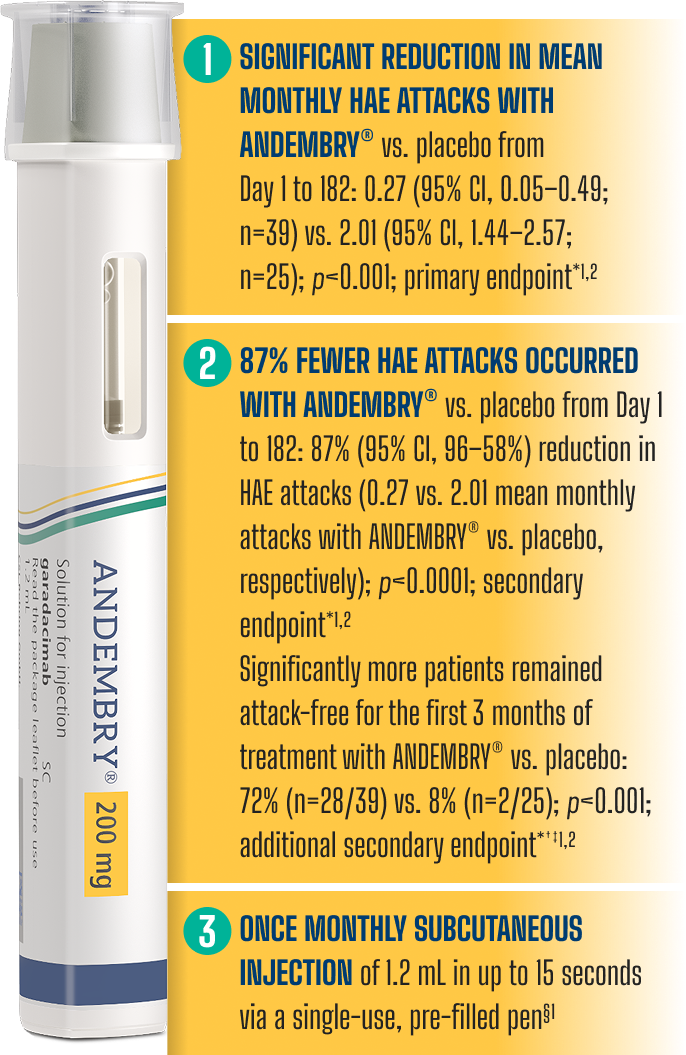
*As demonstrated in the Phase 3 VANGUARD trial: Craig J, et al. Lancet. 2023; 401(10382):1079–1090; ClinicalTrials.gov, NCT04656418.2
† A hierarchical testing procedure controls for the overall alpha level of 5% (2-sided).1
‡ Per the VANGUARD pivotal study, ‘attack-free’ was defined as 100% reduction in attacks vs. baseline, measured during the first and second 3-month treatment periods and over the full 6-month study duration.1,2
§ In Month 1, a loading dose of 400 mg (2x injections) is required.1 ANDEMBRY® may be self-administered only after training by a healthcare professional.1
THE FIRST AND ONLY
FXIIa INHIBITOR FOR HAE1,2
THE FIRST AND ONLY
FXIIa INHIBITOR FOR HAE1,2
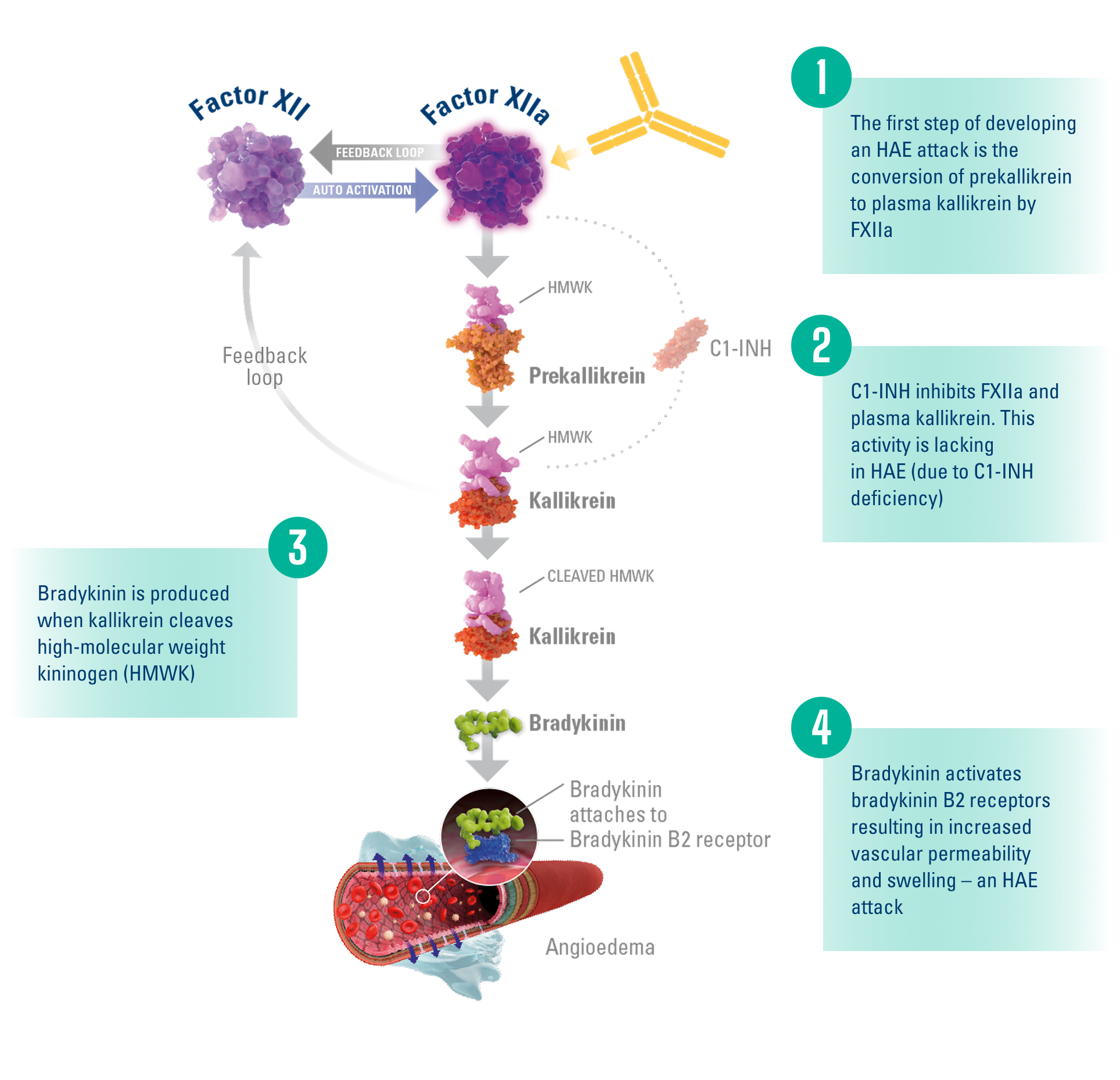
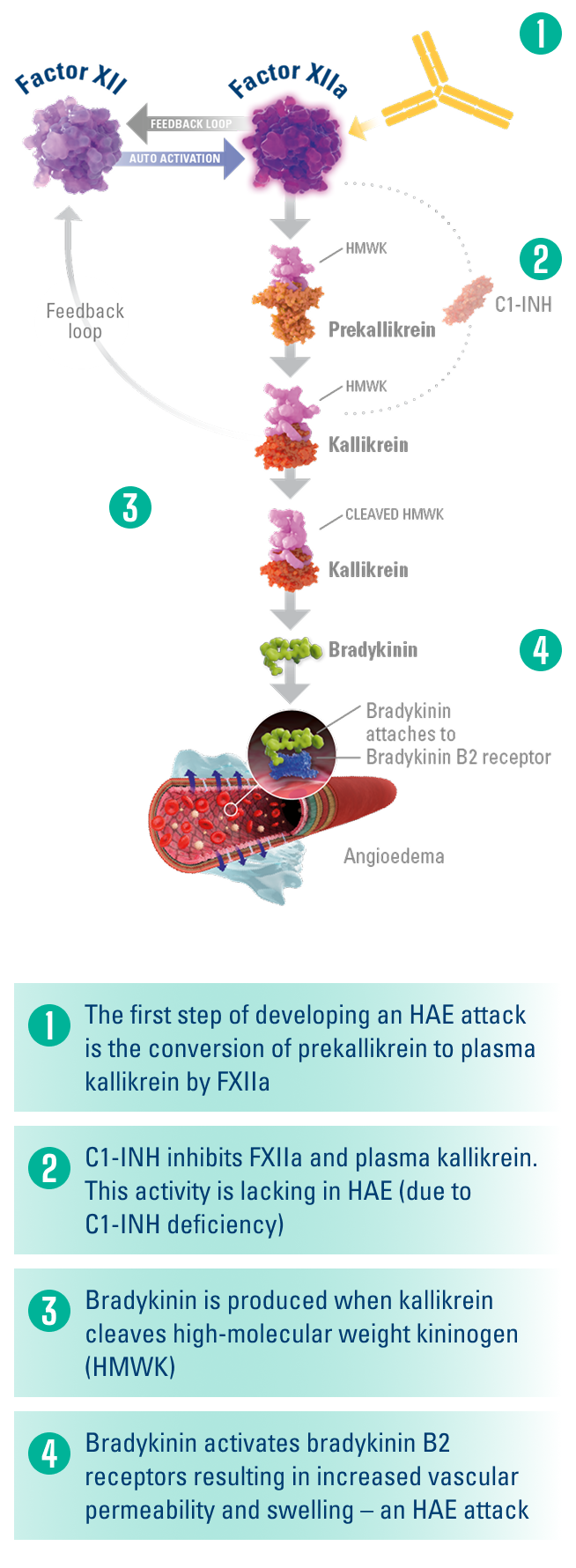
Factor XII is the first protein activated in the contact activation pathway.1,4,5
Factor XIIa plays a key role in HAE attack development by converting prekallikrein to kallikrein, leading to the release of bradykinin.4,5
ANDEMBRY® blocks the function of FXIIa, thereby blocking the conversion of prekallikrein to kallikrein and the subsequent release of bradykinin.1
ANDEMBRY® MoA
In this video, HAE expert Dr Aleena Banerji, MD discusses the MoA of ANDEMBRY® in HAE¶
¶All views are those of the expert speaker and supported by the body of literature summarised at the end of the video.
Simplified HAE Biochemical Pathway4-6
VANGUARD: A PIVOTAL PHASE 3 TRIAL EVALUATING
THE EFFICACY AND SAFETY OF ANDEMBRY®1,2
VANGUARD: A PIVOTAL PHASE 3 TRIAL EVALUATING THE EFFICACY AND SAFETY OF ANDEMBRY®1,2
VANGUARD was a 6-month multicentre, randomised, double-blind, placebo-controlled trial in patients (aged ≥12 years) with type I or type II hereditary angioedema (n=64).¶1,2
VANGUARD study design
In this video, HAE expert and VANGUARD Principal Investigator, Dr Riedl, discusses the study design#
# All views are those of the expert speaker and supported by the body of literature summarised at the end of the video.
STUDY DESIGN¶2,7
01/2021 – 06/20221,2
This was an international Phase 3 study conducted across 7 countries: Canada, Germany, Hungary, Israel, Japan, the Netherlands, and the USA. The study included 58 adults and 6 adolescent patients (≥12 years old).
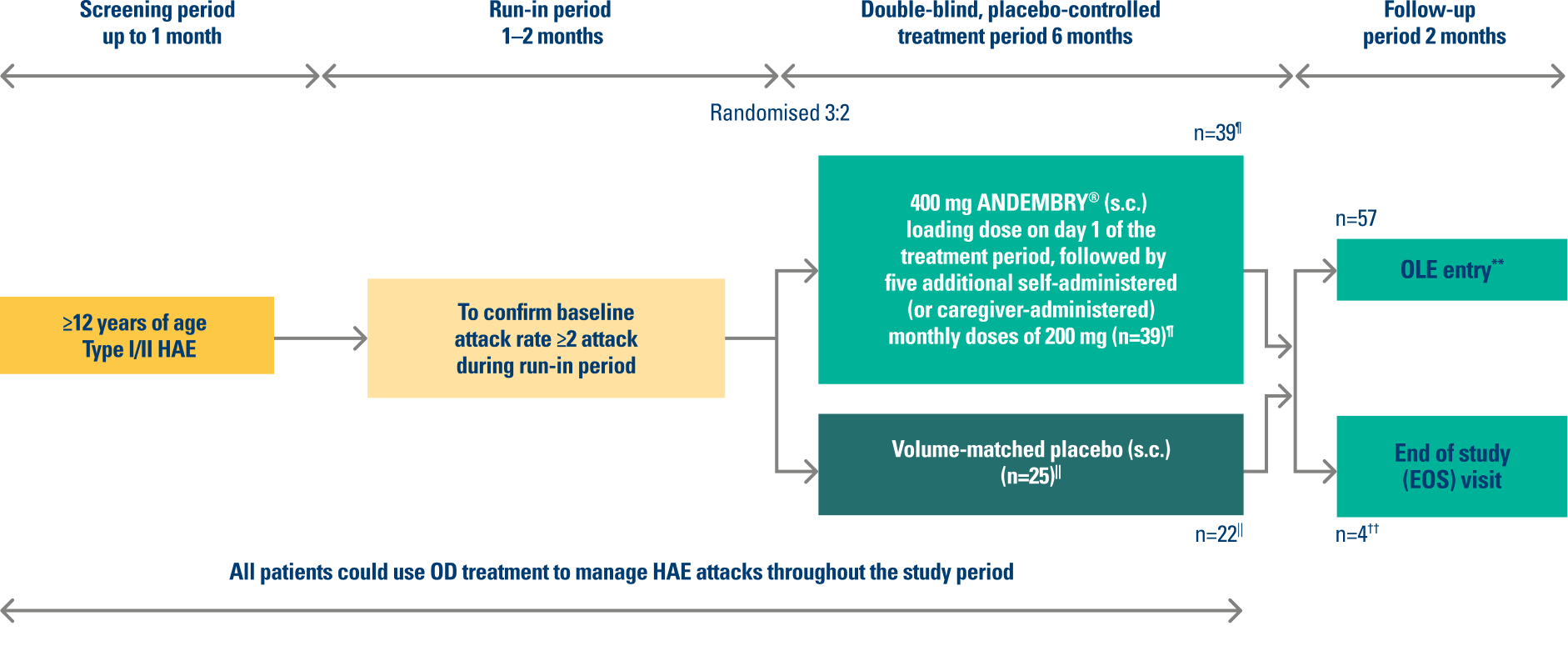
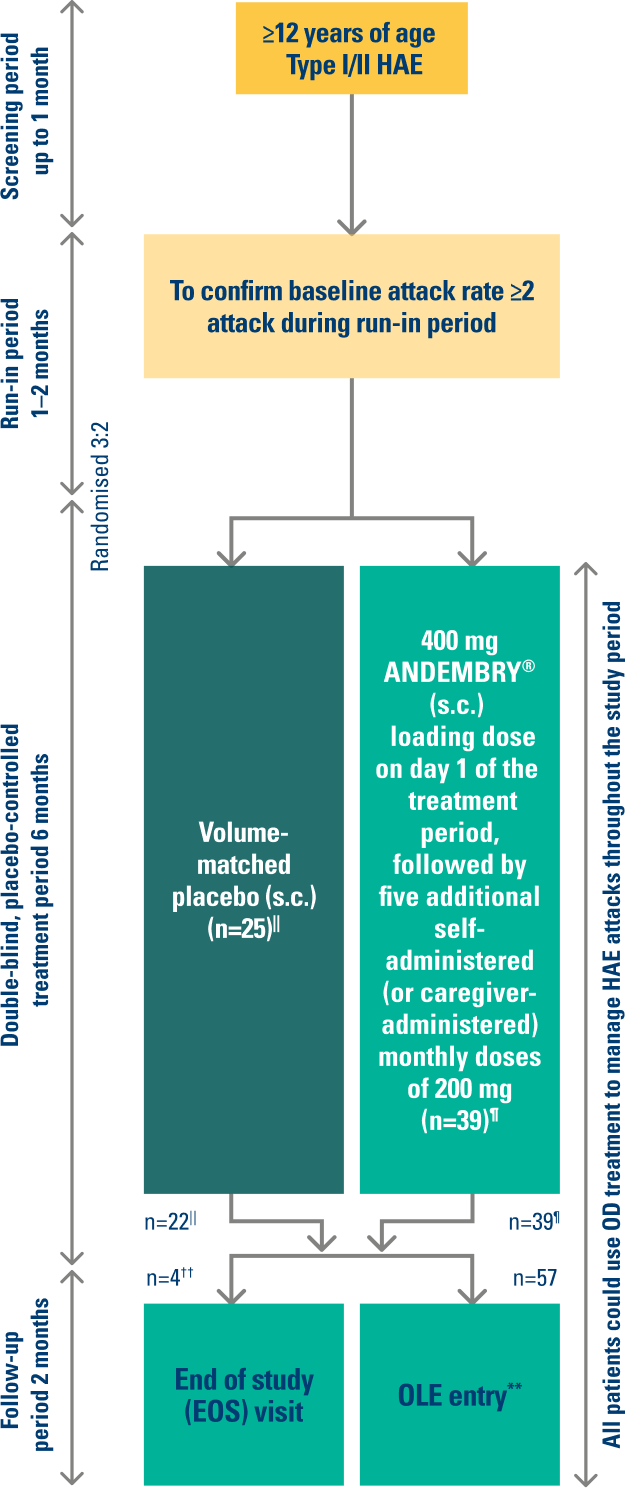
¶ Primary endpoint: investigator-assessed time-normalised number of HAE attacks (number of HAE attacks per month) during the 6-month treatment period (Day 1 to 182). Three secondary endpoints tested in the following hierarchical order: percentage reduction in number of HAE attacks per month vs. placebo (Day 1 to 182); number of attack-free subjects (defined as 100% reduction in attacks vs. baseline) through to Day 91; percentage of patients rating therapy as “good” or “better” with SGART at Day 182. For full patient demographics and inclusion/exclusion criteria, see: Craig J, et al. Lancet. 2023; 401(10382):1079–1090; ClinicalTrials.gov, NCT04656418.2
|| Three patients in placebo arm discontinued the study. One patient discontinued because of lack of efficacy, and two patients withdrew because of HAE attacks.
** OLE study – last patient out November 2025.
†† n=4 patients opted out of the OLE trial for personal reasons.
BASELINE CHARACTERISTICS2
| ANDEMBRY® (n=39) | Placebo (n=25) | Total (n=64) | |
|---|---|---|---|
| Female | 24 (62%) | 14 (56%) | 38 (59%) |
| Male | 15 (38%) | 11 (44%) | 26 (41%) |
| Age at screening, years | 43.3 (17.5) [12–69] | 37.8 (12.8) [14–62] | 41.2 (15.9) [12–69] |
| BMI at screening, kg/m2 | 27.9 (6.0) | 28.4 (7.6) | 28.1 (6.6) |
| Race | |||
| Asian (Japanese) | 4 (10%) | 2 (8%) | 6 (9%) |
| Black or African American | 0 | 1 (4%) | 1 (2%) |
| Native Hawaiian or Other Pacific Islander | 1 (3%) | 0 | 1 (2%) |
| White | 33 (85%) | 22 (88%) | 55 (86%) |
| Other | 1 (3%) | 0 | 1 (2%) |
| HAE type | |||
| Type I | 34 (87%) | 22 (88%) | 56 (88%) |
| Type II | 5 (13%) | 3 (12%) | 8 (13%) |
| Age at diagnosis, years | |||
| ≤17 | 18 (46%) | 12 (48%) | 30 (47%) |
| >17 to ≤40 | 18 (46%) | 11 (44%) | 29 (45%) |
| >40 | 3 (8%) | 2 (8%) | 5 (8%) |
| HAE attacks and treatment history | |||
| Prophylactic therapy during 3 months before screening‡‡ | 14 (36%) | 7 (28%) | 21 (33%) |
| Number of HAE attacks during the 3 months before screening or at the start of prophylaxis | 8.6 (6.3–10.9) | 9.3 (6.4–12.2) | 8.9 (7.1–10.6) |
| Number of HAE attacks during the run-in period | 3.1 (2.4–3.7) | 2.5 (2.1–2.9) | – |
| History of laryngeal attacks | 21 (54%) | 17 (68%) | 38 (59%) |
| Location of HAE attacks during the 3 months before screening§§ | |||
| Cutaneous (extremities) | 30 (77%) | 20 (80%) | 50 (78%) |
| Abdominal | 30 (77%) | 18 (72%) | 48 (75%) |
| Facial | 13 (33%) | 8 (32%) | 21 (33%) |
| Throat, larynx or tongue | 3 (8%) | 2 (8%) | 5 (8%) |
| Peripheral¶¶ | 1 (3%) | 0 | 1 (2%) |
Data are n (%), mean (SD) [range], mean (SD), or mean (95% CI). ANDEMBRY® and placebo were administered every 4 weeks.
‡‡ During the 3 months before entering the run-in period, all 21 (33%) patients receiving HAE prophylaxis discontinued their prophylactic treatments, including C1-esterase inhibitor (subcutaneous or intravenous), berotralstat, lanadelumab, tranexamic acid, and danazol.
§§The full list of primary locations of HAE attacks in the last 3 months before screening is available at: Craig J, et al. Lancet. 2023;401(10382 Suppl.):1079–1090.7
¶¶As described by the investigator using the free text option in the patient’s eDiary.
VANGUARD: ONGOING ASSESSMENT OF
LONG-TERM EFFICACY AND SAFETY OF ANDEMBRY®8
The VANGUARD phase 3 OLE trial aims to evaluate the long-term efficacy and safety of ANDEMBRY® for HAE prophylaxis.8
The trial population includes patients with HAE who participated in the previous phase 2 and 3 trials on ANDEMBRY®, alongside newly enrolled patients.8
Results presented are taken from the latest interim data (cut-off: February 13, 2023; n=161). Median (IQR) treatment exposure was 13.8 months (11.9–16.3 months).8
STUDY DESIGN¶8
03/2021 – 02/2023
This is an international Phase 3 OLE study conducted across 14 countries (Australia, Canada, Czechia, Germany, Hong Kong, Hungary, Israel, Japan, the Netherlands, New Zealand, Russia, Spain, Taiwan, and the USA)
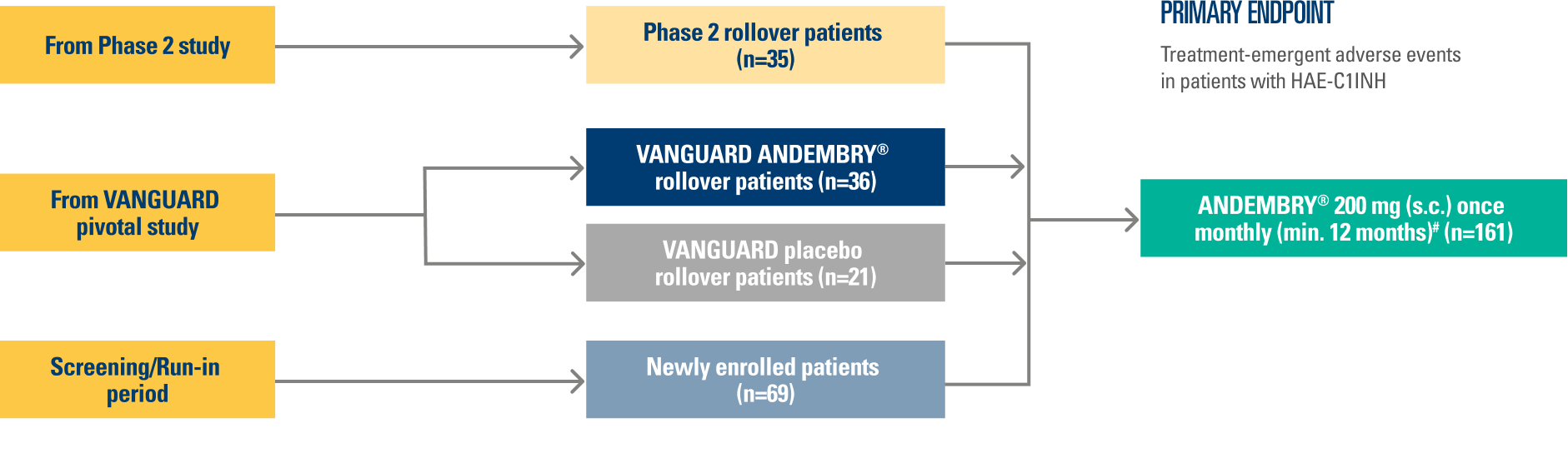
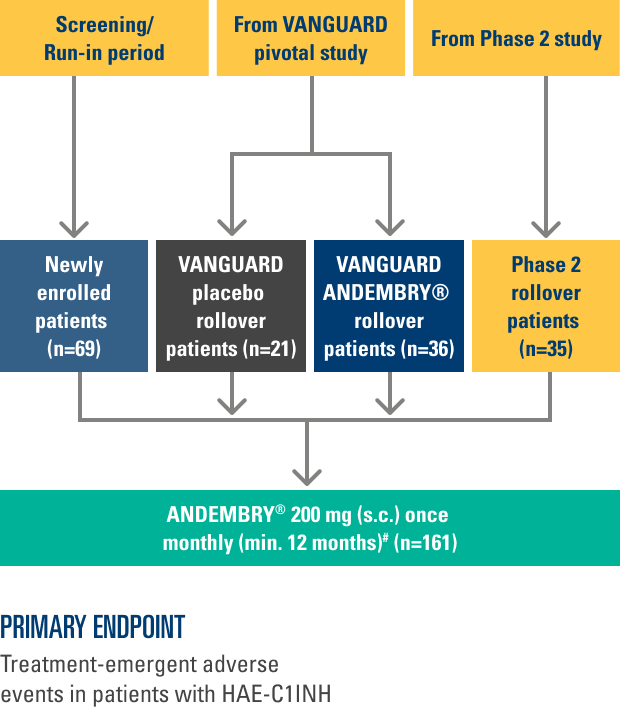
Data presented reflects an interim analysis with a data cut off date of 13 February 2023.
¶The primary endpoint was TEAEs in patients with C1 inhibitor deficiency/dysfunction. For full patient demographics and inclusion/exclusion criteria, see Reshef A, et al. Allergy. 2025; 80:545–556; ClinicalTrials.gov, NCT04739059.8
#Newly enrolled ANDEMBRY® patients were given a loading dose of 400 mg in Month 1.
BASELINE CHARACTERISTICS8
| ANDEMBRY® (n=161) | |
|---|---|
| Female | 101 (62.7%) |
| Mean age at screening, years | 42.3 (15.3) [13–73] |
| Mean BMI at baseline, kg/m2 | 28.1 (6.2) |
| Race | |
| American Indian or Alaska Native | 0 (0%) |
| Asian | 22 (13.7%) |
| Black or African American | 2 (1.2%) |
| Multiple | 1 (0.6%) |
| Native Hawaiian or other Pacific Islander | 0 (0%) |
| Other | 1 (0.6%) |
| White | 135 (83.9%) |
| HAE type | |
| Type I | 145 (90.1%) |
| Type II | 14 (8.7%) |
| HAE-nC1INH | 2 (1.2%) [both with FXII mutation] |
| Age at diagnosis, years | |
| ≤17 | 10 (6.2%) |
| >17 | 151 (93.8%) |
| ≤65 | 148 (91.9%) |
| >65 | 13 (8.1%) |
| HAE attacks and treatment history | |
| History of laryngeal attacks | 103 (64%) |
| Number of HAE attacks during the 3 months before screening or at the start of prophylaxis | 9.3 (8.2–10.4) |
| Number of HAE attacks during the run-in period | 4.7 (4.2–5.1) |
| Patients receiving prophylactic therapy during 3 months before screening | 59 (36.6%) |
| Location of hereditary angioedema attacks during the 3 months before screening | |
| Abdominal | 123 (76.4%) |
| Cutaneous (extremities) | 124 (77%) |
| Facial | 51 (31.7%) |
| Peripheral | 1 (0.6%) |
| Throat | 15 (9.3%) |
| Trunk | 16 (9.9%) |
Explore more

Explore reduced HAE attacks with ANDEMBRY® (VANGUARD)

View the long-term efficacy of ANDEMBRY® (VANGUARD OLE)

Review the safety and tolerability profile of ANDEMBRY®
Abbreviations: BMI, body mass index; CAP, Commercial Access and Pricing; CI, confidence interval; C1-INH, C1-esterase inhibitor; EOS, end of study; FXII, Factor XII; FXIIa, activated Factor XII; HAE, hereditary angioedema; HAE-C1INH, hereditary angioedema with C1 inhibitor deficiency or dysfunction; HAE-nC1INH, hereditary angioedema with normal C1 inhibitor; HMWK, high molecular weight kininogen; IQR, interquartile range; MoA, mechanism of action; NHS, National Health Service; NICE, National Institute for Health and Care Excellence; OD, on-demand; OLE, open-label extension; SD, standard deviation; SGART, Subject's Global Assessment of Response to Therapy; s.c., subcutaneous; TEAE, treatment-emergent adverse events.
References:
1. ANDEMBRY® (garadacimab). Summary of Product Characteristics.
2. Craig J, et al. Lancet. 2023; 401(10382):1079–1090.
3. National Institute for Health and Care Excellence. Garadacimab for preventing recurrent attacks of hereditary angioedema in people 12 years and over (Tec hnology appraisal guidance TA1101) [Internet]. London: NICE; 2025 Oct 8 [cited 2025 Oct 28]. Available from: https://www.nice.org.uk/guidance/ta1101
4. Caccia S, et al. Pediatr Allergy Immunol Pulmonol. 2014; 14(4):159–163.
5. Kaplan AP, Joseph K. Ann Allergy Asthma Immunol. 2010; 104:193–204.
6. Maurer M, et al. Allergy. 2022; 77:1961–1990.
7. Craig J, et al. Lancet. 2023; 401(10382 Suppl.):1079–1090.
8. Reshef A, et al. Allergy. 2025; 80: 545–556.
Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or Apple store. Adverse events should also be reported to CSL Behring UK Ltd. on 01444 447405.
GBR-AND-0213 | May 2026.

