
ANDEMBRY® REDUCED HAE
ATTACKS VS. PLACEBO1,2
ANDEMBRY®
REDUCED HAE
ATTACKS VS.
PLACEBO1,2
ANDEMBRY® (garadacimab) is indicated for routine prevention of recurrent attacks of hereditary angioedema (HAE) in adult and adolescent patients aged 12 years and older. 1
The person represented in this material is not an actual patient.

ANDEMBRY® SIGNIFICANTLY REDUCED MONTHLY
HAE ATTACK RATE VS. PLACEBO1,2
ANDEMBRY® SIGNIFICANTLY REDUCED MONTHLY HAE ATTACK RATE VS. PLACEBO1,2
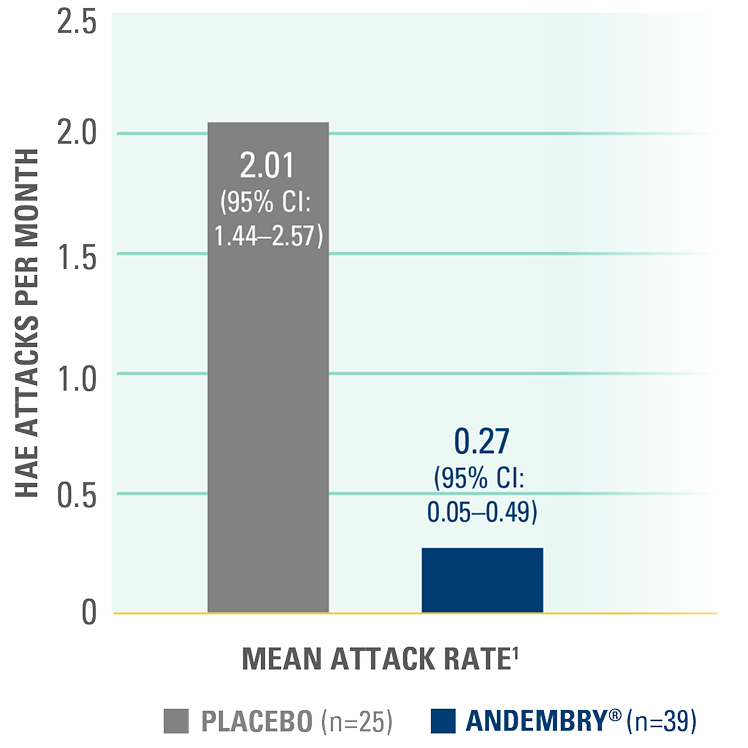
Time-normalised number of HAE attacks (attacks per month) from Day 1 to 182 as assessed by investigators — primary endpoint*1,2
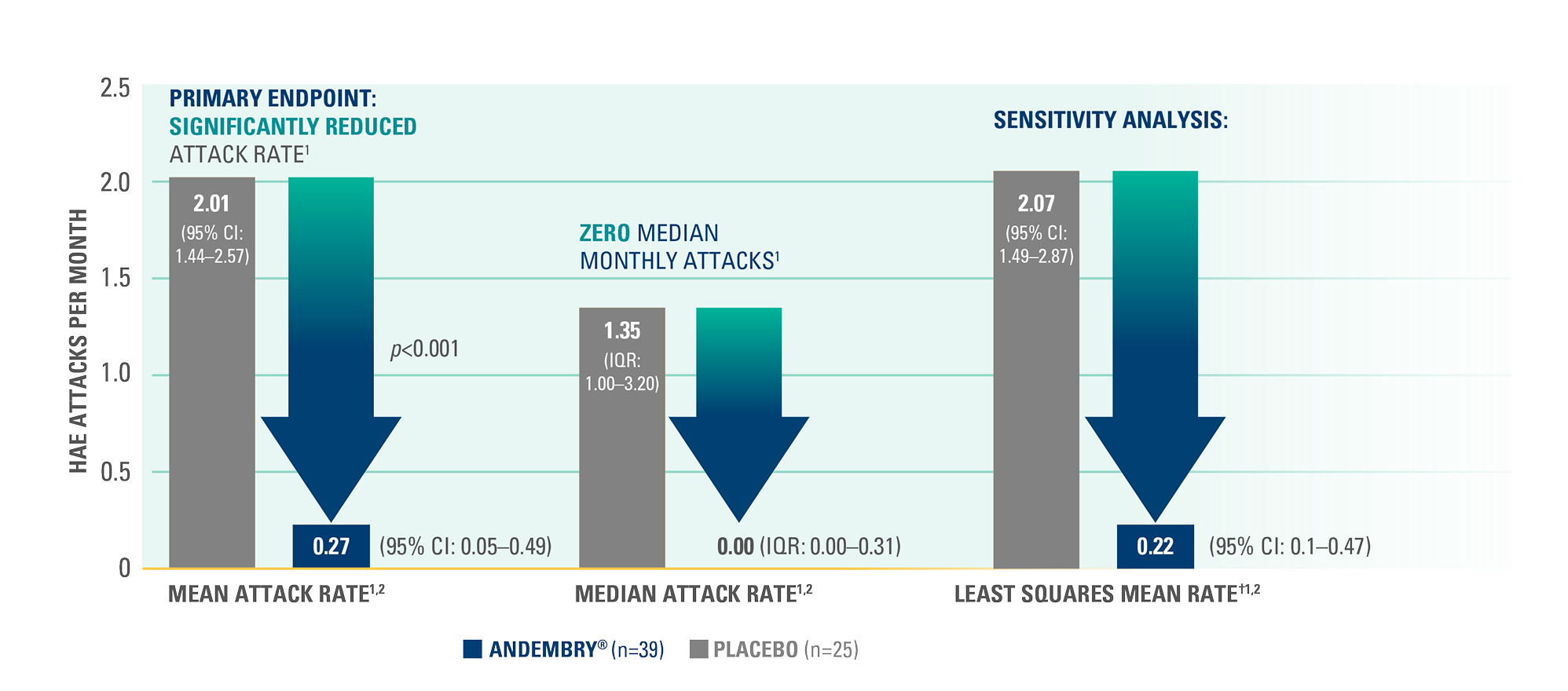
Attack rate per month was significantly reduced with ANDEMBRY® vs. placebo; 0.27 (95% CI, 0.05–0.49) vs. 2.01 (95% CI, 1.44–2.57), respectively; p<0.0011,2
*During the treatment period, 63 vs. 264 HAE attacks occurred with ANDEMBRY® vs. placebo, respectively.1,2
†Baseline-adjusted, time-normalised LSM (sensitivity analysis).1,2
OF PATIENTS TREATED WITH ANDEMBRY®:
87%
87% fewer HAE attacks occurred vs. placebo1,2
72%
72% were attack-free‡ after 3 months1,2
82%
82% reported “Good” or “Excellent” response to therapy1,2
‡Per the VANGUARD pivotal study, ‘attack-free’ was defined as 100% reduction in attacks vs baseline, measured during the first and second 3-month treatment periods and over the full 6-month study duration.
87% FEWER HAE ATTACKS OCCURRED
WITH ANDEMBRY® VS. PLACEBO1,2
87% FEWER HAE ATTACKS OCCURRED WITH ANDEMBRY® VS. PLACEBO1,2
Percentage reduction in the number of HAE attacks per month compared with placebo from Day 1 to 182 — secondary endpoint§1,2
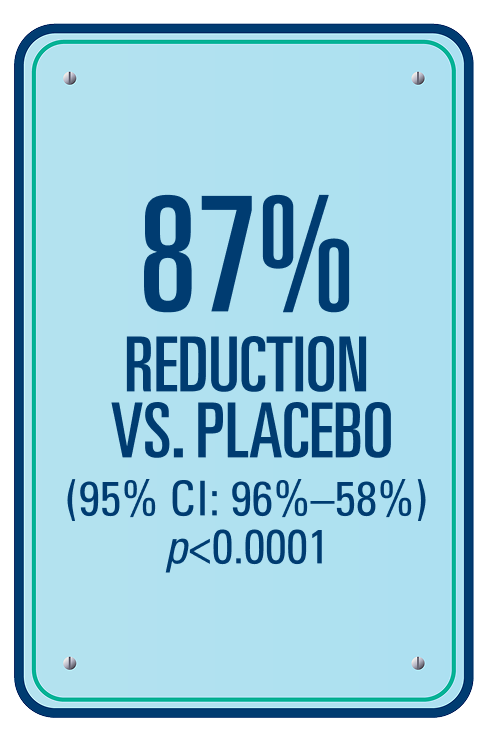
With ANDEMBRY® vs. placebo, there was an 87% (95% CI: 96%–58%) reduction in HAE attacks per month; p<0.00011,2
§A hierarchical testing procedure controls for the overall alpha level of 5% (2-sided).1
72% of ANDEMBRY®-TREATED PATIENTS
WERE ATTACK-FREE‡ THROUGH THE FIRST
3 MONTHS OF TREATMENT1,2
72% of ANDEMBRY®-TREATED PATIENTS WERE ATTACK-FREE‡ THROUGH THE FIRST 3 MONTHS OF TREATMENT1,2
Number of attack-free patients from Day 1 to 91 (end of the first 3-months of treatment) — secondary endpoint‡§1,2
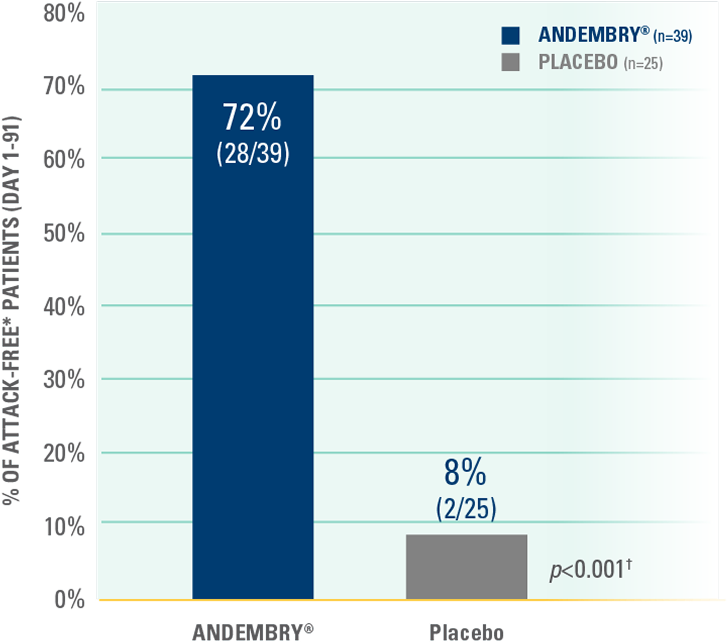
Throughout the first 3 months of treatment, significantly more patients were attack-free with ANDEMBRY® vs. placebo, respectively; 72% (n=28/39) vs. 8% (n=2/25); p<0.001‡§1,2
‡Per the VANGUARD pivotal study, ‘attack-free’ was defined as 100% reduction in attacks vs. baseline, measured during the first and second 3-month treatment periods and over the full 6-month study duration.1,2
§A hierarchical testing procedure controls for the overall alpha level of 5% (2-sided).1
ANDEMBRY® WAS RATED ‘GOOD’ OR ‘EXCELLENT’
BY OVER 80% PATIENTS§1
ANDEMBRY® WAS RATED ‘GOOD’ OR ‘EXCELLENT’ BY OVER 80% PATIENTS§1
Percentage of patients with “good” or “excellent” response to treatment WITH SGART from Day 1 to 182 — secondary endpoint§¶1,2
More patients treated with ANDEMBRY® rated their response to treatment as “Good” or “Excellent” vs. placebo; 82% (n=31/38) vs. 33% (n=8/24), respectively; p<0.001§¶1,2
§The overall response to treatment with the investigational product was self-assessed by the patient using the SGART. Possible responses are ‘none, ‘poor’, ‘fair’, ‘good’ or ‘excellent’.1
¶A hierarchical testing procedure controls for the overall alpha level of 5% (2-sided).1
SGART: Patient
82% (n=31/38)
of patients rated their response to treatment as ‘Good’ or ‘Excellent’ at day 182 vs. 33% (n=8/24) in the placebo group, p<0.0011,2§¶
HEAR FROM AN HAE EXPERT
HEAR FROM AN HAE EXPERT
”In the garadacimab group, 62% of patients were attack-free from the first dose for up to six months compared with zero patients in the placebo group” — Dr Philip Li, MBBS, MD, FRCP, HAE expert and VANGUARD Principal Investigator#
#All views are those of the expert speaker and supported by the body of literature summarised at the end of the video.
VANGUARD efficacy
In this video, Dr Philip Li, MBBS, MD, FRCP, discusses efficacy endpoints from the VANGUARD trial#
WITH ANDEMBRY® THERE WERE...
WITH ANDEMBRY®
THERE WERE...
ZERO
MEDIAN
...MONTHLY ATTACKS requiring rescue medication||1,2
Time-normalised number of HAE attacks requiring on-demand treatment per month (from Day 1 to Day 182) — additional secondary endpoint||1,2
ANDEMBRY® (n=39) median, 0.00 (min. 0.0, max. 3.8) vs. placebo (n=25) 1.35 (min. 0.0, max. 4.4)
Mean (SD), ANDEMBRY® (n=39) 0.23 (0.663) vs. placebo (n=25) 1.86 (1.412)
ZERO
MEDIAN
...MONTHLY moderate or severe attacks||**1,2
Time-normalised number of moderate or severe HAE attacks from Day 1 to Day 182 — additional secondary endpoint||**1,2
ANDEMBRY® (n=39) median, 0.0 (min. 0.0, max. 1.2) vs. placebo (n=25) 0.83 (min. 0.0, max. 4.4)
Mean (SD), ANDEMBRY® (n=39) 0.13 (0.296) vs. placebo (n=25) 1.35 (1.166)
||Non-hierarchically tested endpoints.1,2
**Moderate attacks caused difficulty in performing daily activities or might have required assistance to perform these activities.3 The use of rescue medication was probable.3 Severe attacks caused substantial limitations in the patient’s ability to perform daily activities, might have required medical assistance, and required the use of rescue medication.3
POST-HOC ANALYSIS OF
THE VANGUARD TRIAL
POST-HOC ANALYSIS OF THE VANGUARD TRIAL
TIME-NORMALISED MEAN MONTHLY NUMBER OF HAE ATTACKS AT RUN-IN PERIOD THROUGH TO DAY 182 WITH ANDEMBRY® VS. PLACEBO — POST-HOC ANALYSIS‡‡4
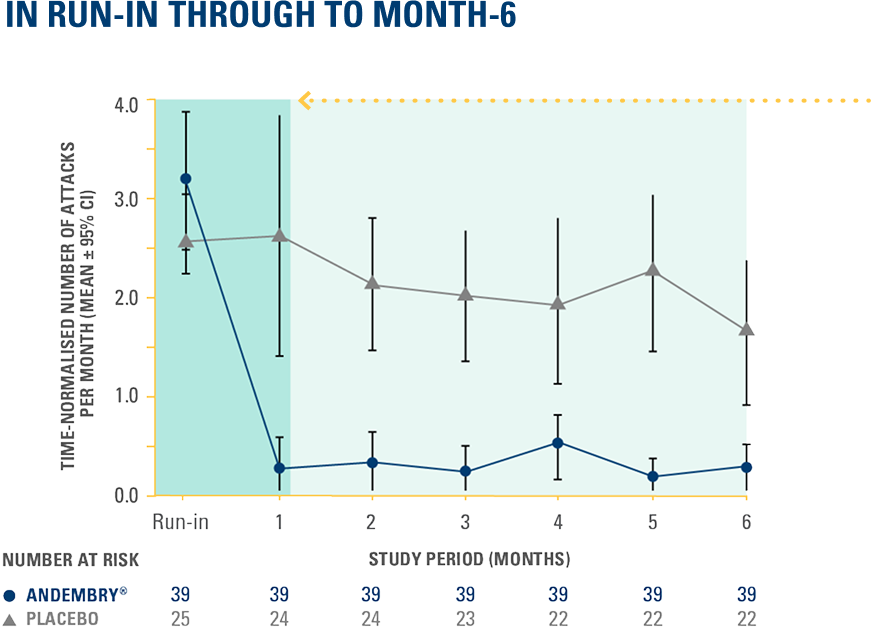
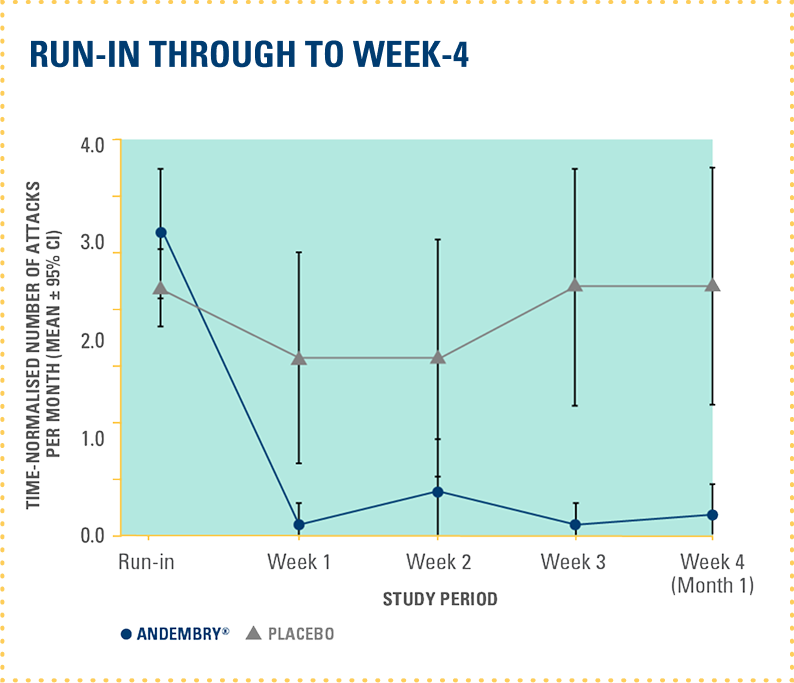
The study did not formally test the statistical hypotheses for post-hoc endpoints‡‡4
‡‡The time-normalised mean monthly number of attacks and percentage of attack-free patients were calculated at weekly intervals for the first 4 weeks and monthly intervals for the rest of the 6-month study duration.4 The onset of protection against HAE attacks was assessed based on actual numbers of attacks (non-extrapolated data).4
Quality of life assessment by AE-QoL domain — exploratory endpoint*1
Changes from baseline greater than the MCID were observed in 88% (n=29/33) of patients treated with ANDEMBRY®#1
*Exploratory endpoints: Data should be interpreted with caution, clinical conclusions cannot be drawn. Further study is required to confirm these results.
#All views are those of the expert speaker and supported by the body of literature summarised at the end of the video.
AE-QoL outcomes in VANGUARD
In this video, Dr Aleena Banerji, MD discusses the impact of HAE on quality of life and AE-QoL endpoints from the VANGUARD trial#
*Exploratory endpoints: Data should be interpreted with caution, clinical conclusions cannot be drawn. Further study is required to confirm these results.
#All views are those of the expert speaker and supported by the body of literature summarised at the end of the video.
CHANGE FROM BASELINE IN AE-QoL QUESTIONNAIRE TOTAL AND DOMAIN SCORES VS. PLACEBO AT DAY 182 — EXPLORATORY ENDPOINT§§1,2
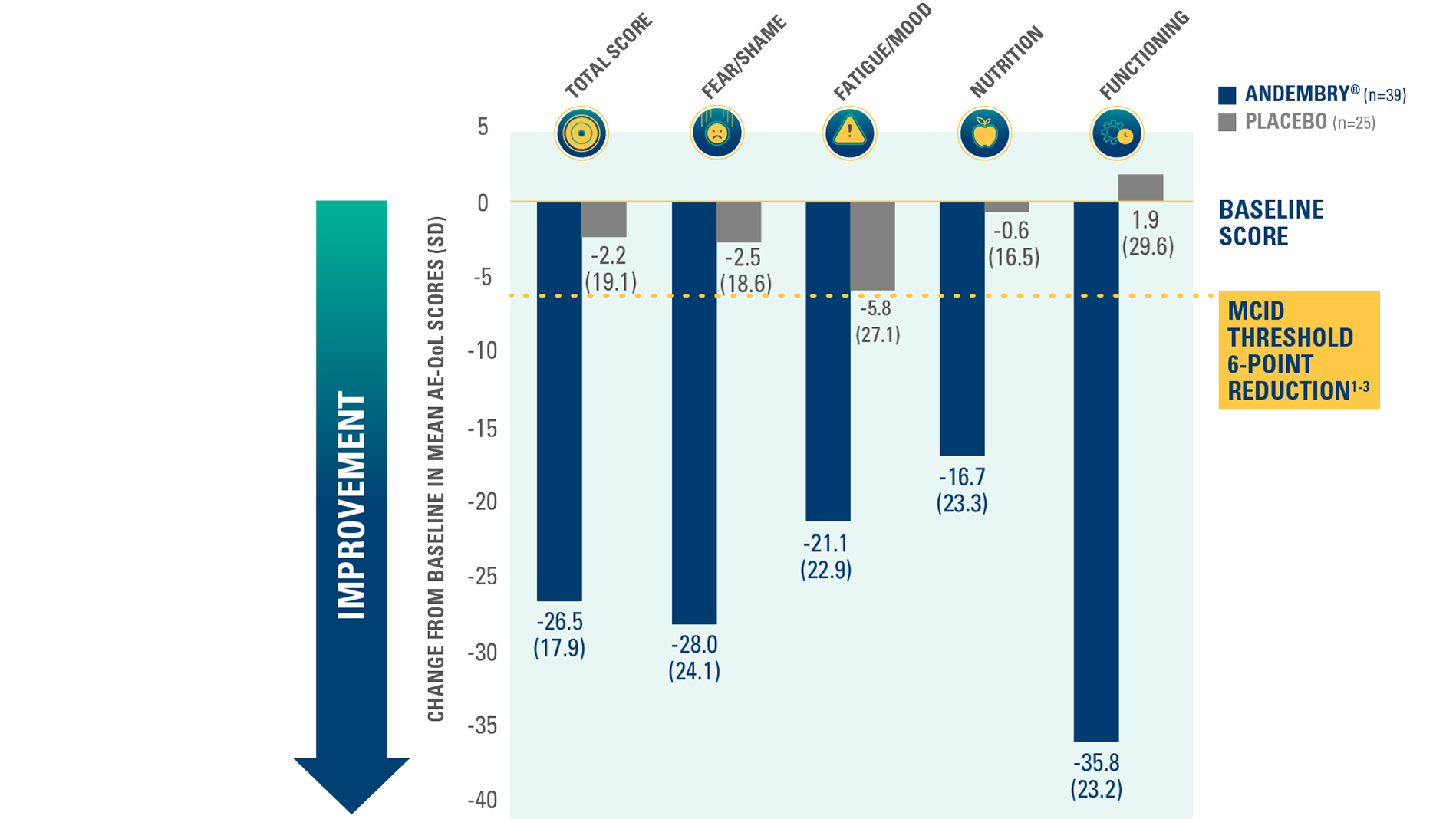
§§A reduction of six points in the AE-QoL has been defined as the MCID.1 AE-QoL was measured in patients of age ≥18 years.1 Lower AE-QoL scores represent greater improvement in quality of life.1
VANGUARD OLE — ATTACK RATES, REDUCTION
AND RESPONSE TO THERAPY5
VANGUARD OLE — ATTACK RATES, REDUCTION AND RESPONSE TO THERAPY5
At the data cut-off for this interim analysis (February 13, 2023), the median treatment exposure (IQR) was 13.8 months (11.86–16.33; range, 3.0–21.1).5
Time-normalised number of HAE attacks per month in the overall population in the run-in and treatment periods — secondary endpoint*¶¶5
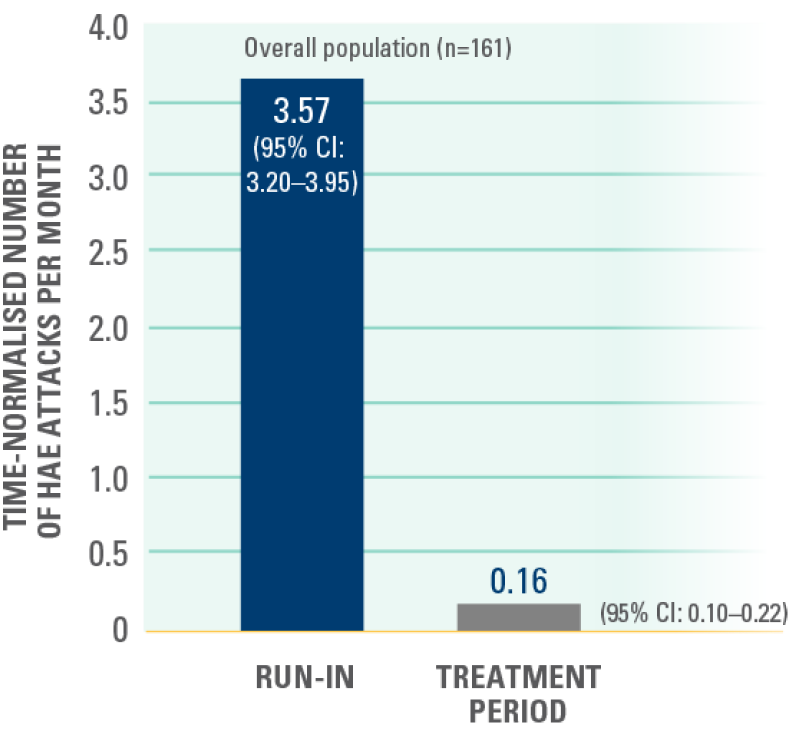

¶¶Statistical analysis not performed.5
Attack-free patients throughout THE treatment period in the overall population — secondary endpoint¶¶5

60% (n=96/161) of patients
remained attack-free with ANDEMBRY®¶¶5
¶¶100% reduction in attack rate.5 Statistical analysis not performed.5
RESPONSE TO THERAPY
93% (n=110/118)
Proportion of patients rating their response to therapy as “Good” or “Excellent” using SGART at Month 12 — secondary endpoint5
98% (n=117/119)
Proportion of patients with “Good” or “Excellent” response to therapy using IGART at Month 12 — exploratory endpoint*5
*Exploratory endpoint: Firm conclusions cannot be drawn from this exploratory endpoint – further study is required.
Explore more

Review the safety and tolerability profile of ANDEMBRY®

Explore how to administer ANDEMBRY®

Discover the latest insights on ANDEMBRY®
Abbreviations: AE-QoL, Angioedema Quality of Life Questionnaire; CI, confidence interval; HAE, hereditary angioedema; IGART, Investigator's Global Assessment of Response to Therapy; IQR, interquartile range; LSM, least-squares mean; MCID, minimal clinically important difference; OLE, open-label extension; SD, standard deviation; SGART, Subject’s Global Assessment of Response to Therapy.
References:
1. ANDEMBRY® (garadacimab). Summary of Product Characteristics.
2. Craig J, et al. Lancet. 2023; 401(10382):1079–1090.
3. Craig J, et al. Lancet. 2023; 401(10382 Suppl.):1079–1090.
4. Staubach P, et al. Clin Exp Allergy. 2024; 0:1–4.
5. Reshef A, et al. Allergy. 2025; 80: 545–556.
Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or Apple store. Adverse events should also be reported to CSL Behring UK Ltd. on 01444 447405.
GBR-AND-0214 | May 2026.
